|
8/14/2023 0 Comments Electron capture chemistry  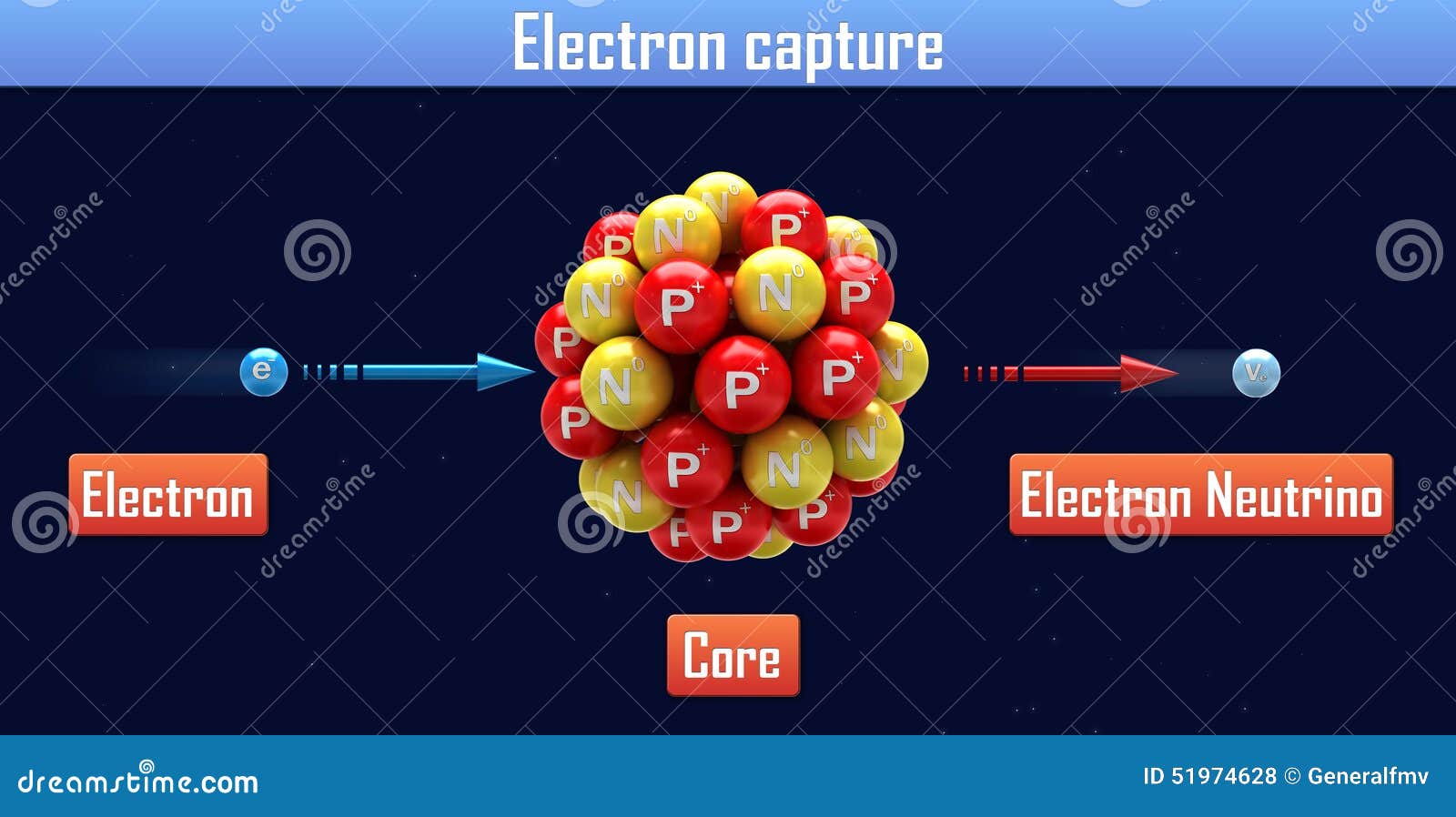
Sensitivity ĭepending on the analyte, an ECD can be 10-1000 times more sensitive than a flame ionization detector (FID), and one million times more sensitive than a thermal conductivity detector (TCD). To laboratory chromatographers this theoretical result is a well known experimental observation. Examination of the rate balance equation with all charge production and loss mechanisms considered reveals that the current collected when the electron capture detector is saturated with analyte is not zero: it is half the current collected when no analyte is present. Negative and positive ions recombine much more rapidly than electrons and positive ions it is this more rapid neutralization that is the origin of the observed decrease in current. The answer is recombination of negative ions or electrons with the positive ions of the makeup gas before these charged entities can be collected at anode and cathode respectively. The key to understanding why the current decreases is to ask where charged entities can go besides being collected at the anode and cathode. It is not immediately obvious why the capture of electrons by electronegative analytes reduces the current that flows between the anode and cathode: the molecular negative ions of the analyte carry the same charge as the electrons that were captured. ECD detectors are particularly sensitive to halogens, organometallic compounds, nitriles, or nitro compounds. Over a wide range of concentrations the rate of electron capture is proportional to the analyte concentration. As the sample is carried into the detector by the carrier gas, electron-absorbing analyte molecules capture electrons and thereby reduce the current between the collector anode and a cathode. There is therefore always a background signal present in the chromatogram. The electrons are accelerated towards a positively charged anode, generating a current. The electrons emitted from the electron emitter collide with the molecules of the makeup gas, resulting in many more free electrons. Usually, nitrogen is used as makeup gas, because it exhibits a low excitation energy, so it is easy to remove an electron from a nitrogen molecule. The electron emitter typically consists of a metal foil holding 10 millicuries (370 M Bq) of the radionuclide 63 The ECD uses a radioactive beta particle (electron) emitter in conjunction with a so-called makeup gas flowing through the detector chamber. The electron capture detector is used for detecting electron-absorbing components (high electronegativity) such as halogenated compounds in the output stream of a gas chromatograph. Electron capture detector, Science History Institute 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
